Cancer reporting tool technical guide
This guidance is associated with:
Cancer incidence in Wales report
Cancer incidence in Wales data
Cancer mortality in Wales report
Cancer mortality in Wales data
Cancer survival in Wales report
Pathology samples indicating new cases of cancer in Wales report
Pathology samples indicating new cases of cancer in Wales data
Introduction
This document provides an overview of the data collection process, data quality and the methodology applied in developing the cancer reporting tool. It also provides definitions, notes for interpretation, and details of where to find further information on cancer statistics in Wales.
In 2009, the Welsh Cancer Intelligence and Surveillance Unit (WCISU) became part of the Health Intelligence Division of the newly created Public Health Wales (PHW). Following a number of structural changes over the years, the Health Intelligence Division has most recently become part of the Research, Data & Digital Directorate (1st October 2024). The WCISU has been responsible for publishing cancer incidence, mortality, and survival statistics in Wales since 1997. Prior to this, such figures were published by the Office for National Statistics (ONS) for both England and Wales.
In accordance with one of the statutory duties of Public Health Wales, the WCISU’s core function is ‘to undertake the systematic collection, analysis and dissemination of information about the health of the people of Wales, in particular cancer incidence, mortality, and survival’. Critical to this duty is the continuous compilation of the national cancer registry of Wales and the production of annual official statistics on cancer incidence, mortality and survival in the resident population of Wales.
The national cancer registry is a live and dynamic database where data is submitted to the WCISU from a range of health care providers and associated services. The WCISU collate and validate the data for each patient, defined as the cancer registration minimum dataset.
Our statistics are produced in accordance with the high professional standards set out in the Code of Practice for Official Statistics. They undergo regular quality assurance reviews to ensure that they meet customer needs and are produced free from any political interference.
A list of the cancer types used in this publication, can be found in Appendix A.
Methodology
Incidence outputs
An incident case of cancer is a newly diagnosed case of:
- A malignant neoplasm: Cancers as described by the World Health Organisation (WHO) International Classification of Diseases as ICD-10 codes C00 to C97
- An in-situ neoplasm: ICD-10 codes D00-D09
- A benign neoplasm: ICD-10 codes D29.2, D32-D33, D35.2, D35.4, D15
- A neoplasm of uncertain or unknown behaviour: ICD-10 codes D37-D48
Note: Not all the above registerable conditions collected by the registry are outlined in the incidence outputs. For example, in 2022 only 61% of registerable cases were categorised as a malignant neoplasm excluding non-melanoma skin cancer (NMSC) – That’s 21,006 cases out of the total 34,531 cases. Secondary tumours and recurrences of a previous cancer are also not counted as new incident cases.
Most cancer sites we report on are categorised based on ICD-10 site codes, which classify cancers according to the anatomical location of the primary tumour. Sites that cannot be accurately defined by anatomical site alone are based on morphology and behaviour codes instead. These include neuroendocrine neoplasms, small cell lung cancers and non-small cell lung cancers. A list of the cancer types reported on in this publication, with accompanying ICD-10 site codes (and ICD-O3 morphology and behaviour codes where applicable), can be found in Appendix A.
Please note that transformations are no longer registered as a separate primary malignant neoplasm from 2018 onwards.
This publication reports on cancers registered in Welsh residents diagnosed between 2002 and 2022 with outputs presented by single and three-year rolling periods for men, women, and persons for:
- Wales
- Health boards
- Local authorities
- Area deprivation fifths for Wales and health boards (three-year periods only at health board level)
Counts, crude rates and European age-standardised rates (EASR) are presented for each of the above breakdowns for all malignant cancer sites and the neuroendocrine neoplasms (Appendix A). Further breakdowns can be found in the data downloads that we haven’t displayed in the tool.
Only counts are provided in the data downloads for the remaining benign, uncertain and in-situ cancer sites (Appendix A).
Cancer incidence by stage at diagnosis is presented for diagnoses between 2011 to 2022 by single and three-year rolling periods for men, women, and persons for:
- Wales
- Area deprivation fifths for Wales
Counts and proportion of cancer diagnosed by stage are presented for each of the above breakdowns for all malignant cancer sites and the neuroendocrine neoplasms where applicable (Appendix A). Staging analysis for leukaemia and brain & central nervous system cancers are not available due to limited information, with analysis for Hodgkin lymphoma, non-Hodgkin lymphoma and myeloma only shown for 2016 onwards due to improvement in staging for these sites in recent years. Consequently, staging analysis for ‘all cancers excluding NMSC’ is not provided. However, staging analysis has been provided for the combined remaining cancer sites and labelled ‘all stageable cancers.’ See Appendix A.
Completeness of staging has improved over the years, so caution is advised when interpreting time series analysis. Due to disclosure issues, we only show all Wales figures by stage and remove cancer sites which have any counts with proportions of 100% for known stages.
Please note that updated analysis has been carried out from 2011 due to rebased population estimates by lower super output area (LSOA) to align with Census 2021 being made available. Analysis of the period 2002-2010 was published on the 26th September 2024 and uses population estimates by LSOA to align with Census 2011.
Mortality outputs
A mortality case of cancer is a death where cancer is recorded as the underlying cause.
A weekly dataset containing deaths registered in Wales residents are supplied from the Office for National Statistics (ONS) for the mortality analysis, where Digital Health & Care Wales (DHCW) provide a weekly snapshot of the data. This was used for the current mortality analysis as opposed to the usual annual dataset as postcodes were needed to link to the latest population figures.
This publication reports on deaths registered in Welsh residents between 2002 and 2024 with outputs presented by single and three-year rolling periods for men, women, and persons for:
- Wales
- Health boards
- Local authorities
- Area deprivation fifths for Wales
Counts, crude rates, and EASRs are presented for each of the above breakdowns for all cancers excluding NMSC, along with 47 other cancer sites. A list of the cancer sites, with accompanying ICD-10 codes, used in this publication can be found in Appendix A.
Survival outputs
Survival analysis includes the first primary neoplasm for each cancer type diagnosed in Welsh residents. For example, if a Welsh patient is diagnosed with a primary malignant breast cancer in 2010 and then a primary malignant lung cancer in 2015, they will be included in the breast survival cohort and the lung survival cohort, but only for their first cancer (i.e. the breast cancer) in the all cancers survival cohort. A list of the cancer types reported on in this publication, with accompanying ICD-10 site codes (and ICD-O3 morphology and behaviour codes where applicable), can be found in Appendix A.
All death-certificate-only (DCO) cases and patients with an invalid date of birth were excluded from the analysis. Patients with zero survival time were included in the analyses, and one day was added to their survival time.
Where possible, one-year, five-year and ten-year net and observed survival (%) (along with 95% confidence intervals (CIs)) are presented by one-year and five-year rolling periods for men, women and persons aged 15-99 (20-99 for bone cancer patients), diagnosed from 2002 to 2022 in Wales for:
- Wales
- Health boards
- Deprivation
- Stage (from 2011 onwards)
Staging analysis for leukaemia, myeloma and brain & central nervous system cancers are not available due to limited information, with analysis for Hodgkin lymphoma and non-Hodgkin lymphoma only shown for 2016 onwards due to improvement in staging for these sites in recent years. Consequently, staging analysis for ‘all cancers excluding NMSC’ is not provided. However, staging analysis has been provided for the combined remaining cancer sites and labelled ‘all stageable cancers.’ See Appendix A.
Completeness of staging has improved over the years, so caution is advised when interpreting time series analysis.
Please note that bladder cancer is only provided for 2007 onwards due to a coding change in that year, and that transformations are no longer registered as a separate primary malignant neoplasm from 2018 onwards. The number of cancer patients at risk, the number of deaths and the number of cases censored for each survival estimate can also be found in the data downloads.
The publicly available survival (Therneau, 2024) and relsurv (Perme and Pavlič, 2018) packages in R 4.4.1 software were used to derive the statistics. Observed survival was estimated using the Kaplan-Meier estimator (Kaplan and Meier, 1958), and net survival was estimated using the Pohar-Perme estimator (Pohar Perme et al., 2012) with the complete approach applied, both capped at 100%. Deprivation life tables for Wales were created in-house to account for background mortality as an approximation to the non-cancer related death rates among cancer patients for net survival (see life tables section). 95% CIs for net survival were estimated using a normal approximation on the survival scale (i.e., the plain method) and were capped between 0 and 100.
The analysis adopted the following post-estimation robustness criteria for each combination of stratifying factors:
- A minimum of 10 patients should be alive at the estimation point being reported (i.e., 10 or more alive one year after diagnosis for one-year survival, 10 or more alive five year after diagnosis for five-year survival, 10 or more alive ten year after diagnosis for ten-year survival)
- A minimum of two deaths should be observed one year either side of the estimation point (i.e., two or more deaths between zero and two years for one-year survival, two or more deaths between four and six years for five-year survival, two or more deaths between nine and 11 years for ten-year survival)
- The standard error at the estimation point should be 20% or less
- The survival estimates should decrease over time (i.e. five-year survival should be less than one-year survival and ten-year less than five-year), otherwise censor five-year/ten-year survival figures
- One-year survival must be present and meet the criteria in order to show five-year survival; one-year and five-year must be present and meet the criteria in order to show ten-year survival
If the above criteria were not met, unstandardised net survival, observed survival and the number of cases, deaths and censored cases were censored for that particular cancer type, period, sex, age group, deprivation quintile/stage/geography level, and survival estimate point (one-year, five-year or ten-year).
One-year, five-year and ten-year age-standardised net survival (along with 95% CIs) were also presented where possible for patients diagnosed aged 15-99 years by one-year and five-year rolling periods, by cancer type and sex at an all-Wales level, using a weighted mean of age-group estimates obtained from the International Cancer Survival Standards (ICSS; Corazziari et al., 2004), Appendix B.
If the robustness criteria mentioned above for unstandardised net survival was not met for each age group, then the two lower age groups (15-44 and 45-54) were combined (15-54, Appendix B) and re-examined. This age specific survival figure was then used for each of the age groups (15-44 and 45-54). Age-standardised net survival, observed survival, and the number of cases, deaths and censored cases were only provided if every age group met the criteria, and these age specific survival figures were also presented. Due to small numbers in younger age groups, only All cancers excl. NMSC, Prostate and Breast cancer are shown as standard groupings, all other cancer sites are combined groupings.
A list of the cancer types along with respective ICD-10 codes used in this publication can be found in Appendix A.
Cancer pathology outputs
This experimental analysis examines the monthly count of individual patients with a pathology sample indicating a primary malignant tumour (excluding NMSC) in Wales for the cancer sites: All cancers, prostate, breast, lung, colorectal and ovary.
Examining monthly cancer pathology samples allows for more timely estimates of cancer incidence in Wales. These new, experimental statistics provide a rapid insight into the impact of the COVID-19 pandemic on pathology-confirmed cases of new cancers, how this impact differed between the cancer types analysed, and a better understanding of post-pandemic recovery of cancer diagnoses.
Cancer registration remains the most authoritative source of data for cancer incidence. However, the time taken to process and validate data by expert cancer registration officers means there is routinely a delay of 18 months before publication (see timeliness and punctuality section). A large proportion of cancer diagnoses are registered using pathology databases, so counts of pathology samples can offer a more timely estimate of cancer incidence.
It is important to note that not all new cases of cancer are verified through pathology and relying solely on pathology information can lead to an underestimation of true cancer incidence compared to cancer registration data. Furthermore, the various methods of cancer detection can be dependent on cancer type, so pathology data may offer more comparable estimates to registration data for certain cancer types compared to other sites. Therefore, caution should be taken when interpreting pathology data.
Data sources
Public Health Wales receives pathology data from the sources listed below:
- Welsh Laboratory Information Management Systems (WLIMS and WLIMS2), hosted by Digital Health and Care Wales (DHCW): For all health boards
- Telepath database: For Cardiff and Vale UHB historic data
Methodology
Table 1 summarises the steps used to process the pathology data, transforming from raw extracts to the final monthly datasets. This demonstrates the percentage of records that are filtered out at different stages of analysis. Note, the percentages shown will vary month by month as the dataset is updated. Table 1 shows that a large proportion of records are filtered out due to factors such as duplicate records i.e. where multiple samples are taken from one patient, and samples that do not match the required definition i.e. non-malignant or non-primary tumours.
The pathology data includes the following information about the tumour in coded form:
- Topography: the site of origin
- Morphology: tumour behaviour
Lookup tables provided by colleagues in Northern Ireland and Scotland cancer registries were used to match topography and morphology codes to ICD-10 codes. Where a match could not be found for a pathology record, that record was excluded.
Individual patients may have more than one pathology sample. A cancer diagnosis may require more than one procedure to indicate cancer; for example, a positive biopsy of the breast and a separate positive dissection of lymph nodes may be the route to diagnose a breast cancer case. Individual patients are therefore only counted once in a two-year period within a specific cancer group (see Appendix C). If a sample is taken that indicates a malignant tumour, the individual cannot be counted again for a sample indicating the same cancer type within two years. However, the same individual can be counted again during this period should a sample indicate a different cancer group. It should be noted that these counting rules differ to those used for cancer registration; cancer registration data only includes primary cancers and does not include recurrences or secondary tumours.
Table 1: Summary of the steps used to process cancer pathology data
Figures as at 09/06/23, covering all data extracted (January 2018 – June 2023)
| % records remaining | |
|---|---|
| Extract data from WLIMS and Telepath | 100% |
| Initial cleaning e.g. removing rows with missing data | 99% |
| Filter out non-primary malignant tumours (behaviour code not 3) | 74% |
| Match topography and morphology codes to ICD-10 codes. Remove all records that could not be matched | 65% |
| Filter out records with an ICD-10 code not beginning with C (these are not malignant tumours) | 61% |
| De-duplicate the dataset | 46% |
| Remove C44 (Non-melanoma skin cancer) | 27% |
Caveats
Cancer pathology is not a substitute for registry data, which remains the most authoritative source of data on new cancers. Pathology data cannot provide a full picture of new cancers because not all new cancers are detected via pathology samples and a sample is not always taken when cancer is diagnosed.
Pathology data therefore provides a more accurate estimate of incidence for some cancers compared to others. The greater the proportion of new cancers detected early by pathology sampling, the more accurate the estimate of incidence will be. Table 2 provides a summary of how pathology data for the cancer sites analysed compared to cancer registration data.
Table 2: Summary comparison of pathology samples vs WCISU cancer registration incidence data, showing the pathology count as a percentage of the registration count. Total counts for the period January 2018 to December 2021
| Cancer type | Pathology samples | WCISU registrations | Percentage (%) |
|---|---|---|---|
| All cancers excluding NMSC | 58652 | 78283 | 74.9 |
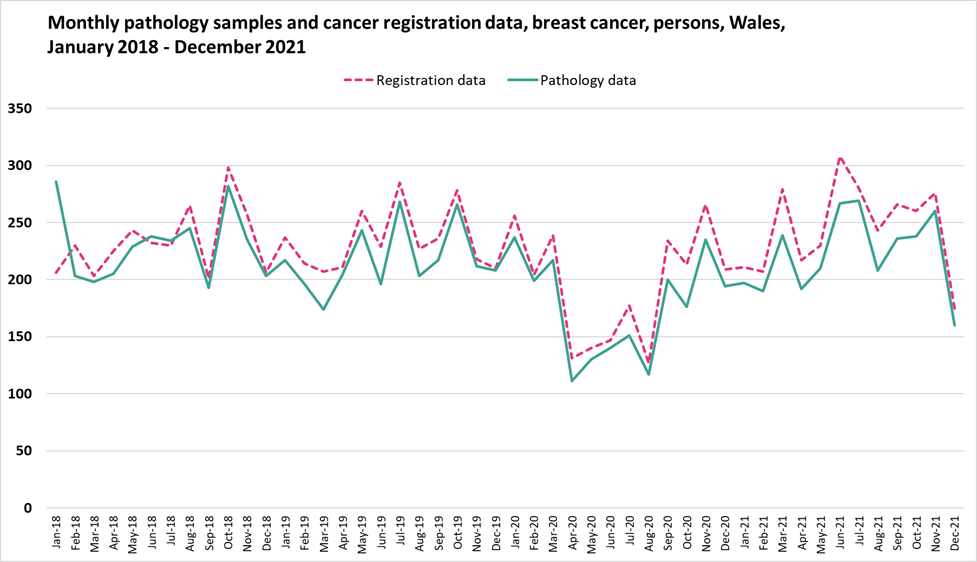
| Breast cancer | 10128 | 10903 | 92.9 |
| Prostate cancer | 6538 | 11203 | 58.4 |
| Lung cancer | 4162 | 9968 | 41.8 |
| Colorectal cancer | 6821 | 9662 | 70.6 |
| Ovarian cancer | 562 | 1297 | 43.3 |
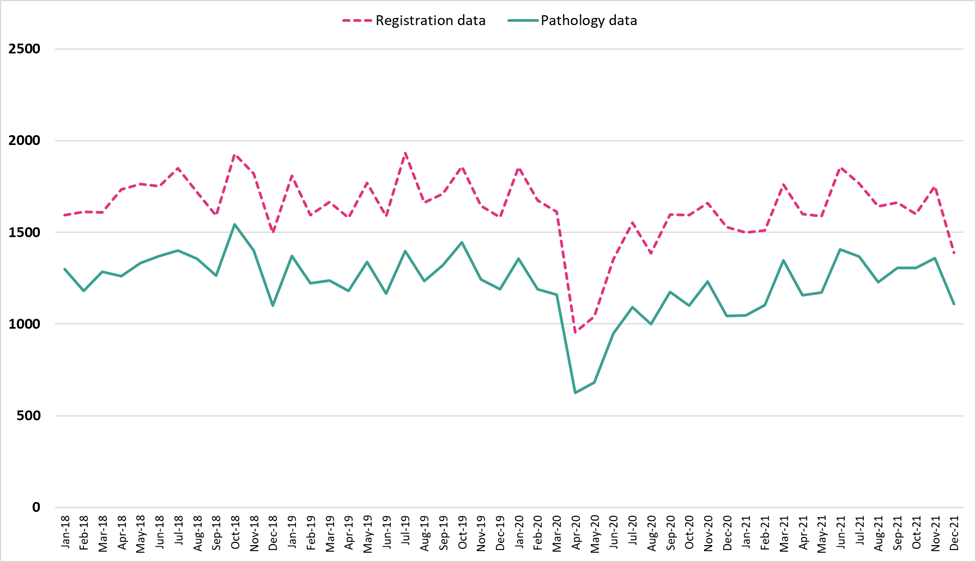
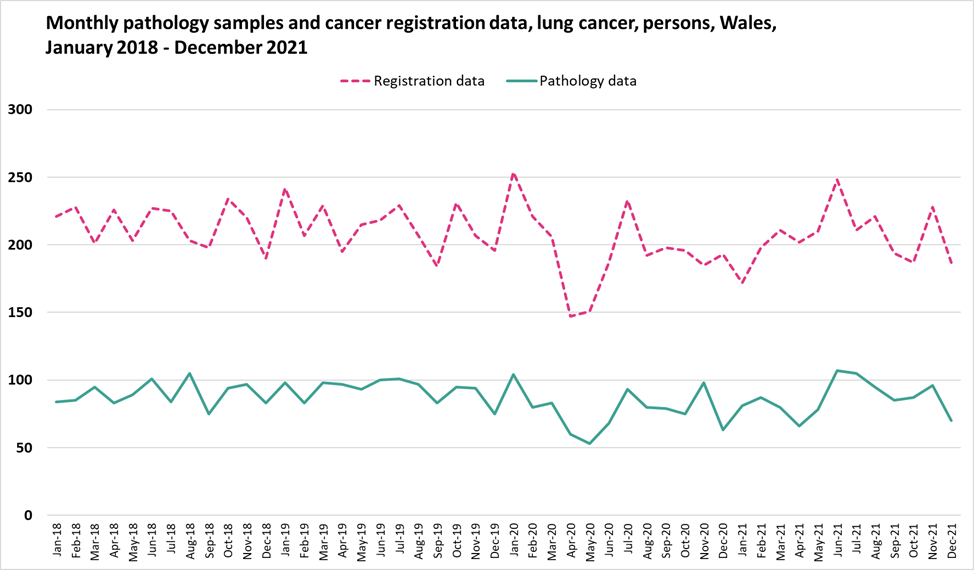
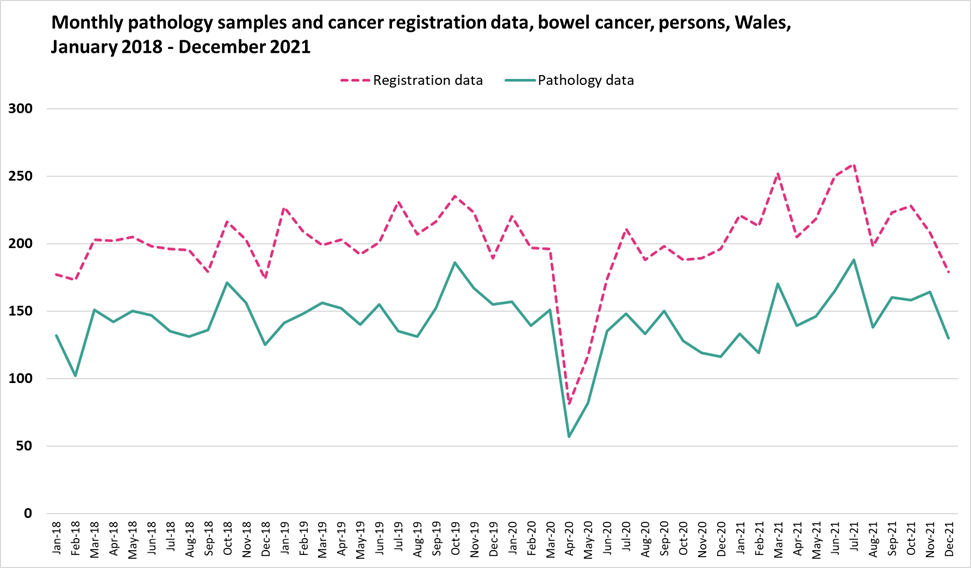
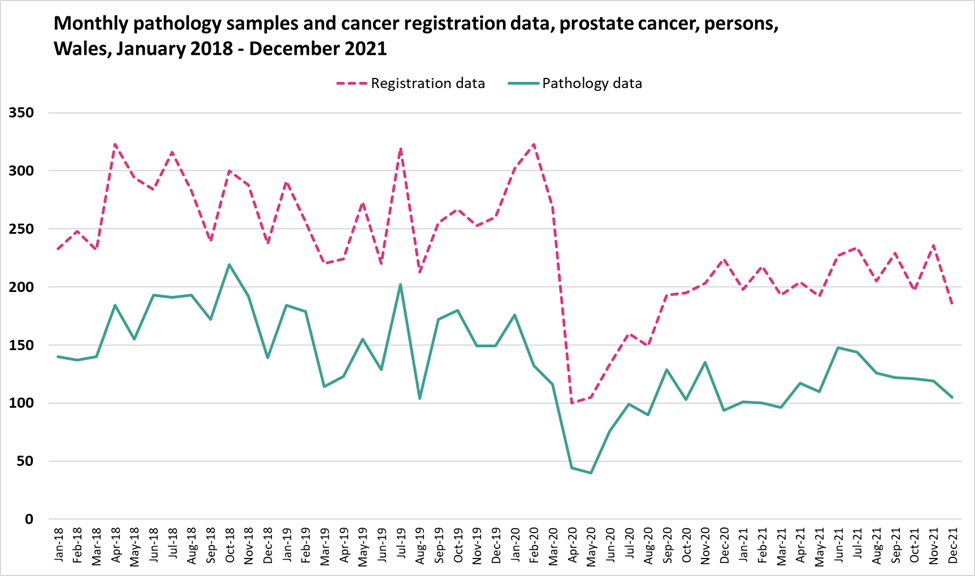
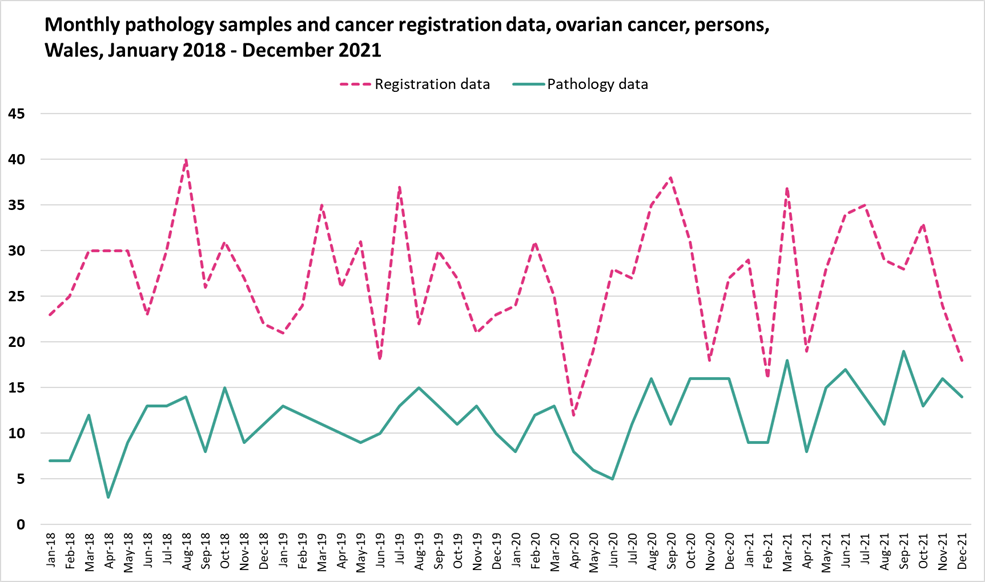
Figure 1 illustrates the monthly pathology data alongside registry data for all cancers excluding NMSC. Pathology samples accounted for around 75% of WCISU cancer registration incident cases for all cancers excluding NMSC. Cancer diagnoses are not always confirmed via pathology tests, so comparability of pathology data to cancer registration data may vary for different cancer sites. See Appendix D for charts illustrating the monthly pathology data alongside registry data for the other cancer sites analysed.
As described above, topography and morphology codes from the pathology data are converted into ICD-10 codes in order to present summarised statistics. There is no single, established lookup table for this conversion process; a bespoke, three-step process was developed using lookup tables provided by colleagues from other UK nations. This process may be adjusted in the future as the method is refined.
On average, of all pathology records with morphology indicating a primary malignant tumour, around 12% each year could not be matched to an ICD-10 code.
The most recent points shown in the chart above are subject to minor changes as the data is updated due to data lags.
Figure 1: Monthly pathology samples vs monthly cancer registration data in Wales for all cancers excluding NMSC, persons, January 2018 to December 2021
Reported characteristics
Geographical area
Analysis presented by geographical area; namely local authority, health board and at an all-Wales level, is based on an individual’s usual area of residence at time of diagnosis for incidence and survival, and at time of death for mortality.
There are seven health boards within Wales. As of 1st April 2019, these are:
- Aneurin Bevan University Health Board
- Betsi Cadwaladr University Health Board
- Cwm Taf Morgannwg University Health Board
- Cardiff and Vale University Health Board
- Hywel Dda University Health Board
- Powys Teaching Health Board
- Swansea Bay University Health Board
Prior to 1st April 2019, the seven health boards were:
- Abertawe Bro Morgannwg University Health Board
- Aneurin Bevan University Health Board
- Betsi Cadwaladr University Health Board
- Cwm Taf University Health Board
- Cardiff and Vale University Health Board
- Hywel Dda University Health Board
- Powys Teaching Health Board
On 1st April 2019 the responsibility for healthcare services in Bridgend County Borough Council area transferred to Cwm Taf UHB (now Cwm Taf Morgannwg UHB) from Abertawe Bro Morgannwg UHB (now Swansea Bay UHB), with the health board boundary moving accordingly. As such, the names of the health boards changed to reflect the new geographical boundaries.
All analyses in this publication are reported using the current health board boundaries, including years prior to 1st April 2019. There are 22 local authority areas in Wales. They were established in 1996 and are nested within the health board areas.
Area deprivation
Analysis by deprivation is presented by deprivation fifths. The Welsh Index of Multiple Deprivation (WIMD) is the official measure of relative deprivation at small area level in Wales. WIMD is made up of eight separate domains of deprivation: income, employment, health, education, housing, access to services, environment, and community safety.
WIMD is used to give an overall deprivation rank for each of the LSOAs in Wales and to provide ranks for the separate deprivation domains for each of the LSOAs.
Deprivation ranks are calculated for each LSOA in Wales. One area has a higher deprivation rank than another if the proportion of people living there that are classed as deprived is higher. The most deprived area is ranked as 1. WIMD is an ecological measure whereas individuals within an area (LSOA in this instance) may vary. Not everyone living in a deprived area is deprived and not all deprived people live in deprived areas. An area itself is not deprived, it is the circumstances and lifestyle of people who are living there that affects its deprivation ranks.
Each of the eight domains are based on a range of different indicators. The domain indices are weighted and combined into an overall index of multiple deprivation.
The deprivation fifths (1 = least deprived; 5 = most deprived) are based on the population, with an equal number of LSOAs in each fifth. Further details on WIMD can be found here.
Analysis based on pre 2011 diagnoses use the WIMD19 to assign a deprivation fifth, whereas analysis based on 2011 diagnoses and onwards, use the WIMD25.
Staging
Great strides have been made to improve the completeness of cancer staging data, allowing cancer by stage in Wales to be presented from 2011 for incidence and survival analysis for malignant cancers.
Stage at diagnosis refers to the size of a tumour and how far it has spread from where it originated. Stage is measured from 1 to 4; stage 1 indicates that the cancer is small and has not spread anywhere, whereas stage 4 indicates the cancer has spread to at least one other body organ. Stage 1 and stage 2 are usually referred to as early stage, whereas stage 3 and stage 4 are referred to as late stage at diagnosis. Cases of cancer recorded on the cancer registry may have an unknown stage for many reasons: patients might be too ill or turn down diagnostic tests to allow clinical staging. In addition, there might be insufficient clinical data received to be able to derive cancer registry stage. Also, a minority of cancer types cannot be staged.
Stage grouping in this report refers to overall TNM stage grouping. The TNM Classification of Malignant Tumours (TNM) is a globally recognised standard for classifying the extent of spread of cancer. T category describes the primary tumour site and size, N category describes the regional lymph node involvement, M category describes the presence or otherwise of distant metastatic spread. From 2018, the majority of cancer types are staged in TNM version 8. Female genital tract cancer also uses FIGO staging which can usually be mapped directly to TNM stage. A change in TNM version can result in a noticeable redistribution between stage groups for some tumour sites, for example, TNM version 7 was introduced in 2016 which resulted in an increase in stage 1 and a reduction in stage 2 for prostate cancers.
In March 2024, changes were implemented in the national cancer registry database to improve the handling of stage fields. The key update was the consolidation of the STAGE_BEST and STAGE_PI_DETAIL fields into a single field for diagnoses 2013 onwards. To ensure consistency in reporting stage across UK publications, STAGE_BEST now only contains stage values for tumours considered stageable by the UKIACR Performance Indicators (PIs). As a result of these changes, a small number of cases that previously appeared staged in groups 1-4 may now appear as stage unknown. Caution is advised when comparing cancer incidence and survival by stage in publications before and after March 2024.
Due to disclosure issues, we only show all Wales figures by stage and remove cancer sites which have any counts with proportions of 100% for known stages.
Completeness of staging has improved over the years, so caution is advised when interpreting time series analysis.
Impact of COVID-19
Due to the Covid-19 pandemic, which began in 2020, additional analysis has been compiled, for all cancers reported on in this publication, detailing how incidence of cancer has changed between 2018-2019 and subsequent years.
This analysis compares the monthly counts of cancer incidence from January to December for the years 2020 onwards for all cancers analysed to an average of the monthly counts for the years 2018 and 2019. Average counts for the months April to December of 2018 to 2019 and April to December of subsequent years are also available; this allows comparison of the period after the first introduction of Covid-19 restrictions, where normal healthcare services were interrupted, to pre-pandemic cancer incidence levels.
Pre-pandemic levels are specified as the average incidence of 2018 and 2019, with the pandemic levels being specified as the incidence during 2020. Comparing these figures provide us with an overview of the impact of the Covid-19 pandemic on cancer patients and cancer services in general.
The impact on net cancer survival in Wales is less straight forward. The life tables produced to account for background mortality use three years of death data and three years of population data. Therefore, because 2020 had unusually high mortality due to COVID 19, any life table that includes this year will be affected. For diagnoses in 2019 and 2021, survival is likely to be slightly overestimated, as the inclusion of 2020 inflates background mortality and therefore increases the number of deaths attributed to non cancer causes. In contrast, survival for diagnoses in 2020 is likely to be slightly underestimated, because the surrounding years (2019 and 2021) dilute the elevated mortality of 2020 and make background mortality appear lower than it truly was. See the life tables section for further information on their use in survival analysis.
Comparisons by United Kingdom (UK) jurisdiction
An additional indicator has been included in the Cancer Reporting Tool allowing comparisons between cancer incidence, mortality and survival data between Wales, England, Scotland, and Northern Ireland. Some cancer types are not comparable as a nation may not report on that cancer type or use different ICD-10 codes to categorise a particular site. Some breakdowns may also be unavailable if there were not enough data points for a useful visualisation within the Cancer Reporting Tool. Only cancer types which are reported by each nation using the same ICD-10 codes are available for comparison. Please be aware that due to differences in coding versions used across different jurisdictions, some sites may not be completely comparable.
Comparisons of cancer incidence and mortality by UK jurisdiction are available for European age-standardised rates and single years, from 2002 until the latest comparable year of data. Cancer survival comparisons by UK jurisdiction are available for net survival (%), and one-year and five-year survival estimates by rolling five-year periods from 2002-2006 until the latest comparable year of data.
Cancer survival is calculated using life tables to account for background mortality (see the life tables section). Life tables are unique to each UK jurisdiction. Therefore, caution is advised when making direct comparisons between Wales, England, Scotland, and Northern Ireland for cancer survival.
For more information on comparability and UK data sources, see the comparability and coherence section. We are currently working with the United Kingdom and Ireland Association of Cancer Registries (UKIACR) to agree a standardised approach to producing life tables, and to categorise cancer types by the same ICD-10 codes to allow more analysis and comparisons between UK nations.
Reported measures
Count
The incidence count is the number of cancers diagnosed over a particular period of time. The mortality count is the number of registered deaths with cancer reported as the underlying cause over a particular period of time.
Crude rates
A crude rate is the number of events occurring in a population over a specific time period, often expressed as the number of events per 100,000 of the population. Both the numerator (number of events) and denominator (mid-year population estimate) are based on the same geographical area and time period.
The crude rate is defined as total registrations per 100,000 population, or:
Crude rates are supressed where there were fewer than three events.
Age-standardised rates
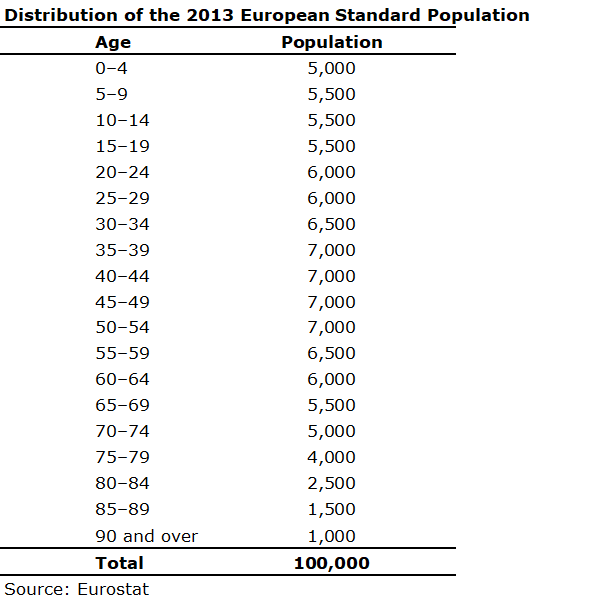
The incidence and mortality of cancer differs greatly with age. Differences in the age structure of populations between geographical areas or over time therefore need to be controlled to give unbiased comparisons. Age-standardisation allows comparison of rates across different populations while taking account of the different age structures of those populations.
This is achieved by applying the observed age- and sex-specific incidence or mortality rates for each population to a standard population. These are then summed to give an overall rate per 100,000 population. The standard population used here is the European Standard Population (ESP) (see Appendix E). The first version of the ESP was introduced in 1976, with the second version in 2013. All the WCISU publications since June 2014 have used the 2013 ESP.
Although the 2013 ESP has separate categories for 90-94 and 95+, the WCISU combines these into a single category for 90+. This is because population data is not consistently available for the 95+ age group for all years or geographies in Wales. The ONS carried out a study looking at the impact of using an 85+, 90+ or 95+ upper age limit for calculating age-standardised mortality rates and found no significant differences between rates calculated with upper age limits of 90+ and 95+. As there is not currently widespread availability of population estimates for the 95+ group, they recommend the use of an upper age limit of 90+ for the 2013 ESP.
A lower age limit of 0-4 years is used for all European age-standardised rates in this tool. This is in line with the other cancer registries within the UK and Ireland.
Age-standardised rates are supressed where there were fewer than 10 events.
The directly standardised mortality rate using the European Standard Population is given by:
where:
- = the observed incidence/mortality rate in age group k
- = registrations in age group k
- = population in age group k
- = 0-4, 5-9, …, 85-89, and 90 and over
- = European standard population in age group k
The 2013 European Standard Population distribution can be found in Appendix E.
Age-specific rates
Age-specific rates are calculated when reporting on cancer incidence and mortality for all sites analysed. An age-specific rate is the number of events occurring in a population over a specific time-period within specific age-groups. Both the numerator (number of events) and denominator (mid-year population estimate) are based on the same geographical area and time period.
The age-specific rate is defined as total registrations per 100,000 population in a specified age group, or:
The age groupings included in the analysis are 0-54, 55-64, 65-74 and 75+.
Proportions
Proportions are calculated when reporting on staging. The sum of all stages should sum to 1 for all cancer sites reported.
Proportions can take values between 0 and 1, and are defined by having:
- A numerator in which every individual known to be in the denominator is counted once or not at all
- A denominator which is exactly defined and known, and in which every individual is counted only once
In such instances, the denominator is considered to be a ‘closed’ population.
Analysis by staging is displayed as percentages in the output, with proportions simply multiplied by 100 to convert to a percentage (%).
Net survival (unstandardised net survival)
Net survival (%) is an estimate of survival where the effect of background population mortality rates on survival has been removed. As background population mortality rates (presented in a life table) are a good approximation to the non-cancer related death rates among cancer patients, the net survival represents the survival of adult cancer patients if they could only die from cancer-related causes. Net survival is suitable for comparison of survival between different time periods and populations, as the confounding effect of non-cancer death rates is removed. However, the analysis is sensitive to differences in background mortality between populations. Please see life tables section for more information about how life tables affect survival estimates. Pohar Perme (2012) detail the net survival method further.
Age specific net survival
Age specific net survival (%) is the same as above but presented by age groups, which are used to calculate age-standardised net survival below. The age groupings used can be seen in Appendix B.
Age-standardised net survival
The survival of cancer varies greatly with age. Differences in the age structure of populations between geographical areas or over time therefore need to be controlled to give unbiased comparisons of survival. Age-standardisation allows comparison of rates across different populations whilst taking account of the different age structures of those populations. Age-standardised net survival (%) is an estimate that would occur if that population (of cancer patients) had an age structure matching that of the general population. Using this metric allows fair comparison of the rates across different regions in Wales, other countries in the UK and Europe, and between different time periods. Survival estimates for five broad age groups are assigned standard weights and summed to give the age-standardised survival estimate (Corazziari et al., 2004). Appendix B details the weights used for the different cancer types.
Observed survival
Observed survival (%) is an estimate of the probability that a patient will be alive at a given time-point after diagnosis (Kaplan and Meier, 1958). It does not take account of cause of death, therefore providing a crude overall prognosis from all possible causes of death. The Kaplan-Meier method is a non-parametric method used to estimate observed survival. For each time interval, observed survival (%) is calculated as the number of subjects surviving divided by the number of patients at risk.
Confidence intervals
Confidence intervals are produced alongside EASRs, age-specific rates and survival figures. They are indications of the natural variation that would be expected around an estimate and should be considered when assessing or interpreting an estimate. The size of the confidence interval is dependent on the number of events occurring and the size of the population from which the events came. Generally, estimates based on small numbers of events and small populations are likely to have wider confidence intervals. Conversely, estimates based on large populations are likely to have narrower confidence intervals.
In this publication, we calculate 95 per cent confidence intervals. This represents a range of values that we can be 95 per cent confident contains the ‘true’ underlying estimate.
Confidence intervals calculated alongside EASRs use the modified gamma distribution method by Tiwari, R.C. et al. (2006). This is a modification of the formula for the upper confidence limit of the original confidence intervals for directly standardised rates based on the gamma distribution, as proposed by Fay and Feuer (1997). Confidence intervals are presented alongside proportions using a method proposed by Wilson, E.B. et al. (1927).
Comparisons are often made between two or more estimates, for example, between different areas or time periods (Figure 2). Sometimes in such cases statistical testing is undertaken by comparing the confidence intervals of the estimates to see if they overlap. Non-overlapping confidence intervals are considered as statistically significantly different. Whilst it is safe to assume that non-overlapping confidence intervals indicate a statistically significant difference, it is not always the case that overlapping confidence intervals do not. A more exact approach is to calculate the ratio of the two estimates, or the difference between them, and construct a test or confidence interval based on that statistic. Such methods are not covered in this technical guide but can be found in a standard textbook.
Figure 2: Using confidence intervals for making comparisons
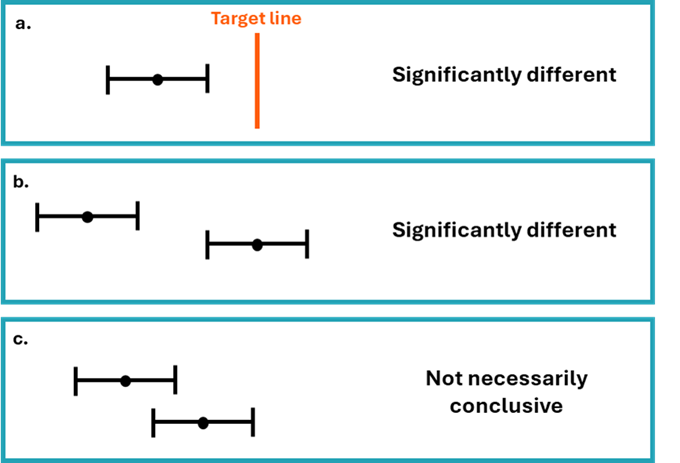
Geographical analysis in this tool indicates whether the EASRs or net survival is significantly different compared to the Wales rate for the area, cancer site and time-period. In this instance, significant difference is indicated by whether the confidence intervals for a particular area overlap or not with the confidence intervals around the Wales estimate for the cancer site and time-period.
Populations
ONS mid-year population estimates from the relevant time periods are used as the denominator for rate calculations. Full guidance on the methodology used by ONS to calculate population estimates can be accessed here.
All recent publications use rebased population estimates by LSOA from mid-2011 to the most current reported year as the denominator for the geographical analysis to align with Census 2021. Details on rebasing of mid-year estimates can be accessed here. Analysis presented for 2002-2010 uses population estimates by LSOA as the denominator for the geographical analysis to align with Census 2011.
For mortality, population estimates by LSOAs for mid-2023 and mid-2024 were not available at the time of analysis therefore 2022 populations were used as a proxy for 2023 and 2024 for the analysis.
Datasets and analysis
All analyses were carried out using reproducible analytical pipelines in R. These analyses were carried out under the following versions:
- Incidence analysis – R 4.4.0
- Mortality analysis – R 4.2.2
- Survival analysis – R 4.4.1
The interactive profile, ‘Cancer Reporting Tool,’ was created using RShiny version 2022.07.2+576 and published via shinyapps.io.
Data snapshots are taken and analysed for each Official Statistics publication produced by the WCISU.
The snapshot of the cancer registration database for survival statistics was taken in July 2025 and followed up until 5th April 2025.
The snapshot of the cancer registration database for incidence statistics covers data from 2011 onwards, while a frozen dataset is used for the years 2002–2010 (taken in June 2024) due to differences in geographic boundaries over time. The snapshot for the latest cancer incidence publication was taken in July 2025.
For mortality statistics, a snapshot from the weekly dataset (taken 19th May 2025) containing deaths registered in Wales residents supplied from the Office for National Statistics (ONS) were used – where Digital Health & Care Wales (DHCW) provide the weekly snapshots of the data. This was to enable linkage to rebased Census 21 populations. Previously, an annual ‘frozen’ dataset was used.
Relevance
The release of Official Statistics by the WCISU is authorised in law by:
- The Pre-release Access to Official Statistics (Wales) Order 2009
- Official Statistics (Wales) Order 2013 made under section 65(7) of the Statistics and Registration Service Act 2007
We believe the key users of statistics regarding cancer incidence, mortality and survival are:
- The public and community groups
- NHS Wales as a whole, and Health Boards, Trusts, and the NHS Wales Cancer Implementation Group and Wales Cancer Network, as well as other teams in Public Health Wales and other national and local public bodies
- Professional bodies, clinicians of all disciplines, and policy makers
- Cabinet secretary, Ministers, and their civil servants
- Other government departments
- Senedd Members and the Members Research Service
- Third sector and charities
- Media
- Students, academics, and universities – to provide valuable reference data for academics and researchers to engage in cancer related research, including the WCISU’s direct participation in research collaborations
- The private sector
We encourage users of our Official Statistics to contact us to let us know how they use the data. Please see the contact details at the top of this page. Acknowledgement to Public Health Wales NHS Trust is to be stated if material in this document or accompanying outputs is reproduced.
Accuracy
Cancer registration
The registration of cancer cases is a dynamic process in the sense that the database is always open and changing. The database is dynamic in a number of ways:
- New cancer cases will be registered: this can include new ‘late’ registrations, where a case is registered after the cancer registry have published what were thought at the time to be virtually complete results for a particular year
- Cancer records can be amended: for example, the site code would be modified should later and more accurate information become available
- Cancer records can be deleted, although this is relatively unusual
In common with cancer registries in other countries, cancer registrations in Wales can take up to five years after the end of a given calendar year to reach 100% completeness, due to the continuing accrual of late registrations, amendments, and deletions.
Wales implemented a new cancer registration system named CATRIN in 2015. This is the same as the ENCORE system used in NHS England (formally known as Public Health England), which has inbuilt registration validations. This modernisation programme has improved cross border data sharing in particular. The data migration process placed a particular emphasis on reducing duplicate registrations existing in both Wales and England registry databases. A quality assurance and de-duplication exercise was undertaken to rationalise the cancer registrations across the two countries. Therefore, this may reflect in a reduction in incidence.
Data is supplied by multiple data providers from NHS Wales Health Boards and Trusts, DHCW (formally known as NHS Wales Informatics Service), NHS England, and elsewhere within Public Health Wales, such as its Screening Division. The national cancer registry of Wales is a live and dynamic database of cancer incidence data from 1972 onwards, with approximately 20,000 patients diagnosed each year in Wales (excluding non-melanoma skin cancer).
Cancer registrations comply with a quality assurance framework comprising of a suite of quality checks performed at various time points during the registration year e.g., monthly, quarterly and end of year checks. These check the data consistency of the cancer site, sex and associated histology as well as validity checks on dates, for example, to check invalid combinations for behaviour and site/histology; check that the incidence date is not after the date of death. These checks align to those published in 2018 by the European Network of Cancer Registries (ENCR).
All our outputs include information on coverage, timing, and geography.
Once the expected cancer records for any registration year have been validated, a snapshot of the data is taken to ensure that there is a consistent set of data behind the official statistics for a period of 12 months. Subsequent snapshots of data are taken monthly and can be used in further cancer publications, queries, and parliamentary questions. For information on the snapshots used in our most current Official Statistics publications, please see the datasets and analysis section.
When the WCISU submits registrations for the next reporting year, they can also submit ‘late registrations’ for previous years. If any new ‘late’ registrations for earlier years passed all quality checks, they would be included in the subsequent refreshed dataset. This results in small differences in the underlying number of cancer registrations for previous reports, although these changes are unlikely to have a meaningful impact on cancer incidence. In the unlikely event of incorrect data being published, revisions would be made, and users informed in conjunction with the Code of Practice.
As the data comes from different sources, the quality and accuracy of the data submitted may vary.
ICD-10 coding system changes
ICD-10 coding for cancer is based on the nature and anatomical site of the cancer. Previous Official Statistics publications mapped to and reported on ICD-10v0 for all diagnosing years. However, publications from 2022 onwards will report on ICD-10v0 for diagnosing years 2001 to 2012, and ICD-10v4 from 2013 onwards.
There have been changes and revisions regarding the coding of some cancers using ICD10v4, such as the introduction of new diagnosis codes along with minor changes to some cancer descriptions and naming conventions. There have been some more significant changes related to the coding of blood cancers, mainly non-Hodgkin lymphoma and leukaemias being coded more precisely. Therefore, caution is advised when examining trends for these sites as well as sites of unknown or unspecified origin. The change in reporting of ICD-10 from 2013 onwards should also be taken into consideration when examining data.
Morphology and behaviour codes
Morphology and behaviour codes describe the characteristics and type of cancer cells and how they behave. From 2016 onwards, the WCISU have coded morphology in ICD-O3. Prior to this, coding used ICD-O2. A mapping has been developed and applied to cases pre-2016 in order to determine the equivalent ICD-O3 morphology for consistency. A small number of cases have not been able to be mapped. We are investigating these cases and developing tools for their inclusion in future.
ONS data
It is a legal requirement to register a death and so ONS mortality data provides a reliable and complete data source.
There have been two recent revisions to the way the death certificates are translated by the Office for National Statistics into International Classification of Diseases codes (10th revision). These changes mean that unrevised data are not comparable across years. The main change relates to the rules that govern which cause of death detailed on the death certificate is selected as the underlying cause. Comparability ratios have not been used in these analyses and therefore caution should be exercised when interpreting trends. See the datasets and analysis section for information on the data used for this year’s publication.
Life tables
Survival analysis estimates the net survival (%) of patients with cancer. These survival figures have been used to produce unstandardised and age-standardised estimates. The model uses population life tables to remove background mortality, and assumes the remaining mortality is due to cancer.
Net survival analysis involves comparing the survival of patients with cancer with background mortality (the survival of the general population). For reasons of practicality, our background mortality data includes cancer deaths. This is unlikely to skew our net survival figures for specific cancer sites, but it would likely have an effect on deaths from all cancers, because cancer accounts for about a quarter of all deaths. For that reason, caution is advised when interpreting survival for all cancers combined, and focus should be on the change over time as opposed to the survival estimate.
The life tables were developed in-house and modelled using a flexible Poisson model. The modelled life tables use three years of death data and three years of population data centred around the year of interest in analysis. This methodology was based on the Rachet et al. (2015) paper and a Standard Operating Procedure (SOP) is currently being produced by the United Kingdom and Ireland Association of Cancer Registries (UKIACR).
Methodology
Overarching methodology
In this analysis, the methodology for constructing life tables has been refined to include deprivation quintile as a covariate in the flexible Poisson model, with an interaction effect between age and deprivation. This represents a change to last year’s approach, where separate models were fitted for each combination of year, sex, and deprivation fifth. The current approach allows for the borrowing of information across sub-groups, which is particularly beneficial for groups with small population sizes that may otherwise exhibit volatile mortality rate estimates. The inclusion of an age * deprivation interaction term allows the effect of deprivation on mortality to vary with age. This approach ensures that the model captures the complex relationship between deprivation and mortality across the life course.
Modelling covariates
For the main effect of age on mortality, the best-fitting cubic spline form of age was selected based on the Akaike Information Criterion (AIC) and visual assessments of model fit. The model considered 7-, 8-, and 9-knot solutions, with some knots fixed at key ages and others determined through simulation of multiple possible configurations.
For the age * deprivation interaction term, we assessed models using a simple linear form of age, as well as 3- and 4-knot cubic splines. The best-fitting approach was selected using the AIC.
Separate models were applied for each year and sex combination to account for temporal and sex-specific differences in mortality.
Handling of mortality data for ages 90+
In the dataset used for modelling (which contains raw population and death counts stratified by year, sex, deprivation quintile, and age), mortality data for individuals over 90 years is not provided in single-year increments. Instead, the total number of deaths for all individuals aged 90+ is supplied, while population estimates remain available for each individual year of age.
To address this, we estimate a median age of mortality for individuals over 90 and apply the aggregated 90+ mortality rate to this specific age. Since our model extrapolates trends in deprivation-specific mortality beyond this median age, and given that deprivation inequalities in mortality tend to diminish at older ages, this approach can sometimes result in a crossover effect in death rates at the upper age range (approximately ages 93–99). To mitigate this, the model including deprivation was restricted to age 89, and from age 90 onwards a model including only age was fitted. The resulting age-specific rate of change was then applied to each deprivation fifth from their age 89 mortality rates, effectively fixing deprivation inequalities at age 89.
The effect of Covid-19 may be seen for diagnosis periods including 2019 onwards for one-year survival, 2015 onwards for five-year survival and 2010 onwards for ten-year survival. This analysis uses modelled life tables developed in-house which do not fully account for changes in background mortality due to the Covid-19 pandemic.
Due to data availability for small area populations at the time of publication, cancer patients diagnosed in 2002 use the 2002-2004 lifetable. As a result of using three-year rolling average lifetables, survival for diagnoses in 2019 and 2021 are likely to be slightly overestimated (as background mortality was higher in 2020 due to the pandemic) and survival for diagnoses in 2020 is likely to be underestimated (as 2019 and 2021 deaths form part of the background mortality for this cohort). We are currently considering methods to adjust for these issues and intend to implement these in future analyses. However, it is important to acknowledge that consistency in methodology is important for comparisons across and between years, so the three-year rolling periods for life tables have been maintained.
Timeliness and punctuality
All outputs adhere to the Code of Practice by pre-announcing the date of publication through the upcoming calendar on the Welsh Government Statistics and research page. Furthermore, if publication needs to be postponed this will be announced and the reason for the change fully explained, as set out in the Code of Practice.
Incidence and survival
Historically, the WCISU has routinely published data on incidence and survival within 18 months of the end of the calendar year with a survival publication following. The lapse in time is due to the source data being completed and becoming available, the time taken to process and merge all cancer registrations for those patients’ resident in Wales into one record for each tumour using the data sources provided to the WCISU, according to strict international rules and guidelines of coding, classification and staging.
Accessibility and clarity
The statistics will be published in an accessible, orderly, pre-announced manner on the Welsh Cancer Intelligence and Surveillance Unit’s website at 9:30am on the day of publication. We also publicise the outputs on social media and to our stakeholders via email. All outputs are available and free to download.
Official Statistics are sent to several individuals on the pre-release list five working days prior to the announcement in accordance with the Pre-publication Official Statistics Order Access (Wales) 2009. The individuals on the pre-release list can be found on the publication webpage.
We aim to use plain English in our outputs, and they adhere to the Public Health Wales’s accessibility policy. Furthermore, all our statistics are published in Welsh and English. Further information regarding the statistics can be obtained by emailing [email protected].
Comparability and coherence
While the WCISU does not hold cancer data about residents in Northern Ireland, Scotland, and England, comparable data can be located from the following:
- Northern Ireland Cancer Registry (NICR)
- Public Health Scotland (PHS)
- Office for National Statistics (ONS)
- National Disease Registration Service (NDRS)
Details of cancer registries in the United Kingdom and Ireland can be found on the United Kingdom and Ireland Association of Cancer Registries website.
For comparable incidence and mortality data, the latest international comparisons are available from Cancer in Five Continents.
Due to differences in the life tables between different cancer registries, we advise against making direct comparisons between the different UK Jurisdictions. We are working with other registries towards improving comparability. In the interim, the European/world studies are suitable for comparisons between the different UK Jurisdictions as the same methodology was applied, however data for recent periods is not yet available (Eurocare).
The WCISU have recently published their 2021 UKIACR performance indicators. Further information regarding the data completeness and quality of cancer registry data is detailed in the UKIACR performance indicators reports.
Legislation
Under the Data Protection Act, the lawful processing of patient/service user data for purposes other than that necessary for the direct provision of care requires one of the following conditions to be met:
- Explicit patient/service user consent for processing
- Explicit authorisation by statute
- Approval under Section 251 of the NHS Act 2006
Public Health Wales undertakes a number of activities that cannot be classed as direct care, but where the obtaining explicit consent would be impractical or would compromise the integrity of the relevant activity. Examples include:
- Evaluation of screening programmes
- Cancer registration
- Registration of congenital anomalies
So called ‘Section 251’ approval, therefore, remains the most appropriate means of ensuring that Public Health Wales complies with the Data Protection Act when undertaking such processing. Such approval needs to be obtained for new activities and renewed annually for existing activities.
Section 251 was established to provide a secure legal basis for the disclosure and processing of confidential information in the NHS where it is not possible to use anonymised information or to obtain explicit consent. A mechanism was established to enable the Secretary of State for Health to exercise powers of approval under Section 251, advised by the National Information Governance Board (NIGB) and its Ethics and Confidentiality Committee (ECC). The mechanisms operated by NIGB and its ECC also applied to Wales.
Under General Data Protection Regulation (GDPR), we follow:
- Article 6 (1) e – processing is necessary for the performance of a task carried out in the public interest or in the exercise of official authority vested in the controller
- Article 9 (2) h – processing is necessary for the purposes of preventive or occupational medicine, for the assessment of the working capacity of the employee, medical diagnosis, the provision of health or social care or treatment or the management of health or social care systems and services on the basis of Union or Member State law or pursuant to contract with a health professional and subject to the conditions and safeguards referred to in paragraph 3.
The key policy on cancer in Wales is set out in the Wales Cancer Network’s Cancer improvement plan 2023-2026.
The Well-being of Future Generations Act 2015 is about improving the social, economic, environmental, and cultural well-being of Wales. The Act puts in place seven well-being goals for Wales. These are for a more equal, prosperous, resilient, healthier, and globally responsible Wales, with cohesive communities and a vibrant culture and thriving Welsh language. Under section (10) (1) of the Act, the Welsh Ministers must:
- publish indicators (“national indicators”) that must be applied for the purpose of measuring progress towards the achievement of the Well-being goals
- lay a copy of the national indicators before the National Assembly. The 46 national indicators were laid in March 2016
Further information on the Well-being of Future Generations (Wales) Act 2015.
The statistics included in this release could also provide supporting narrative to the national indicators and be used by public services boards in relation to their local well-being assessments and local well-being plans.
The WCISU adheres to the United Kingdom and Ireland Association of Cancer Registries (UKIACR) small numbers guidance for cancer incidence, i.e. where a cancer incidence count of less than five is observed in any cell with the population at risk being less than 1,000 then the value is suppressed. WCISU also adheres to the Office for National Statistics disclosure control guidance for mortality statistics.
Glossary
- ASR – Age-specific rate
- DHCW – Digital Health & Care Wales
- EASR – European age-standardised rate
- ECC – Ethics and Confidentiality Committee
- ENCR – Eurpoean Network of Cancer Registries
- ESP – European standard population
- FIGO – International Federation of Gynaecology and Obstetrics
- GDPR – General Data Protection Regulation
- HB – Health board
- ICD – International Classification of Diseases
- ICSS – International Classification of Standard Weights
- LA – Local authority
- LSOA – Lower super output area
- MYE – Mid-year population estimates
- NHS – National Health Service
- NDRS – National Disease Registration Service
- NICR – Northern Ireland Cancer Registry
- NIGB – National Information Governance Board
- NMSC – Non-melanoma skin cancer
- ONS – Office for National Statistics
- PHS – Public Health Scotland
- PHW – Public Health Wales
- UHB – University Health Board
- UKIACR – United Kingdom and Ireland Association of Cancer Registries
- WCISU – Welsh Cancer Intelligence and Surveillance Unit
- WHO – World Health Organisation
- WIMD – Welsh Index of Multiple Deprivation
- WLIMS – Welsh Laboratory Information Management Systems
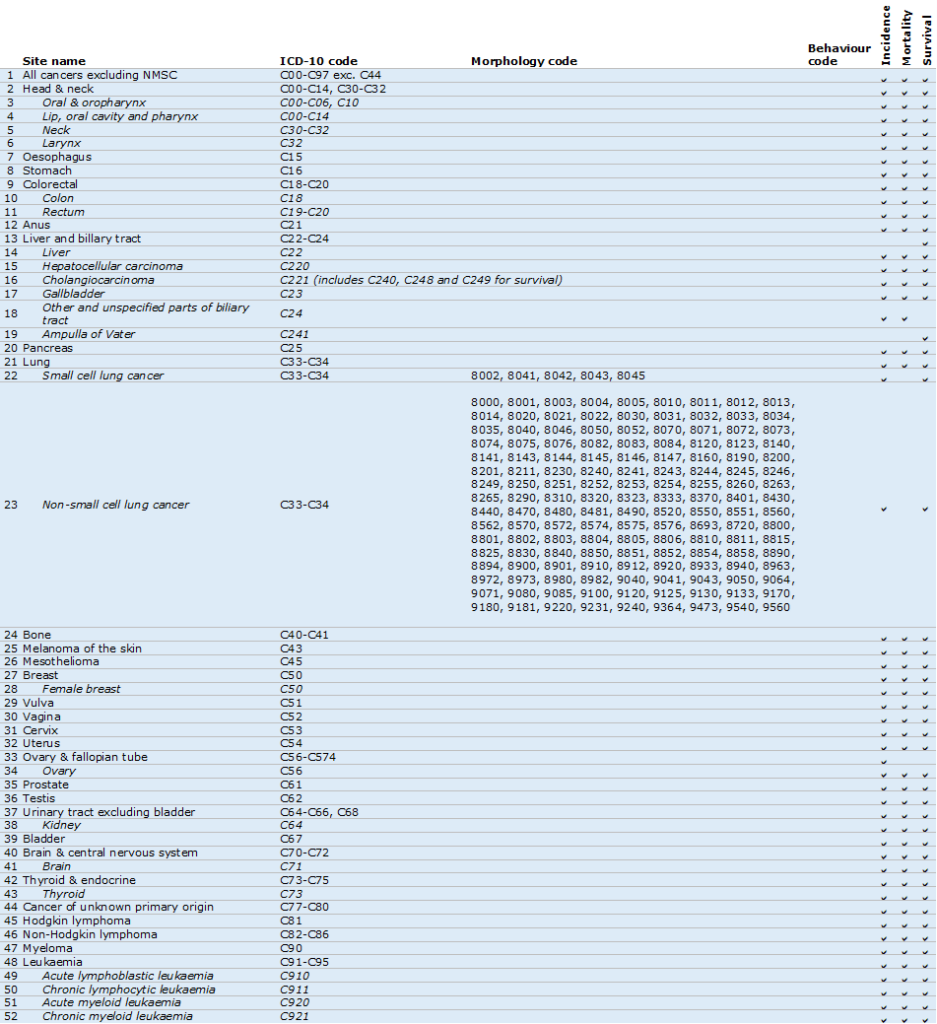
Appendix A
Cancer type map 1: Malignant cancers
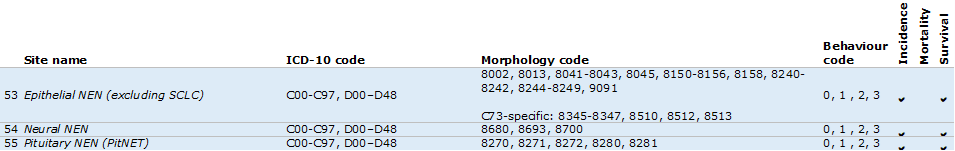
Cancer type map 2: Neuroendocrine neoplasms (NEN) (also includes small cell lung cancer in above table)
Cancer type map 3: Benign and uncertain cancers
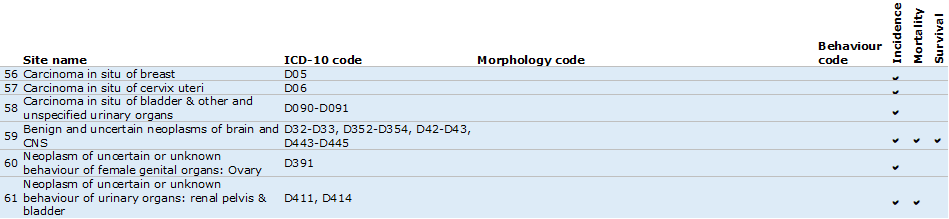
Cancer type map 4: Stageable cancers

Appendix B
International Classification of Standard Weights (ICSS)
| Age band | ICSS1 | ICSS2 | ICSS3 | Age band | Prostate |
|---|---|---|---|---|---|
| 15-44 years | 7000 | 28000 | 60000 | 15-54 years | 19000 |
| 45-54 years | 12000 | 17000 | 10000 | 55-64 years | 23000 |
| 55-64 years | 23000 | 21000 | 10000 | 65-74 years | 29000 |
| 65-74 years | 29000 | 20000 | 10000 | 75-84 years | 23478 |
| 75-99 years | 29000 | 14000 | 10000 | 85-99 years | 5522 |
| 15-99 years | 100000 | 100000 | 100000 | 15-99 years | 100000 |
ICSS1: Lip, tongue, salivary glands, oral cavity, oropharynx, hypopharynx, head & neck, oesophagus, stomach, small intestine, colon, rectum, liver, biliary tract, pancreas, nasal cavities, larynx, lung, pleura, breast, corpus uteri, ovary, vagina & vulva, penis, bladder, kidney, choroid melanoma, non-Hodgkin lymphomas, multiple myeloma, chronic lymphatic leukaemia, acute myeloid leukaemia, chronic myeloid leukaemia, leukaemia, all cancers
ICSS2: Nasopharynx, soft tissues, melanoma, cervix uteri, brain, thyroid gland, bone
For bone cancers, Corazziari et al. (2004) recommend using ages 20+ for survival and so ICSS2 with lower age band 20-44 has been used.
ICSS3: Testis, Hodgkin lymphoma, acute lymphatic leukaemia
Note: On some occasions the age standardised survival rate for some cancer types and diagnosis periods will be slightly higher or lower for persons compared with men and women individually. This is due to the survival rates by age band and the ICSS weights used to calculate the age standardised rates. It should be noted that the age standardised survival rate for persons will be a better survival estimate compared to men and women individually due to the smaller 95% confidence interval.
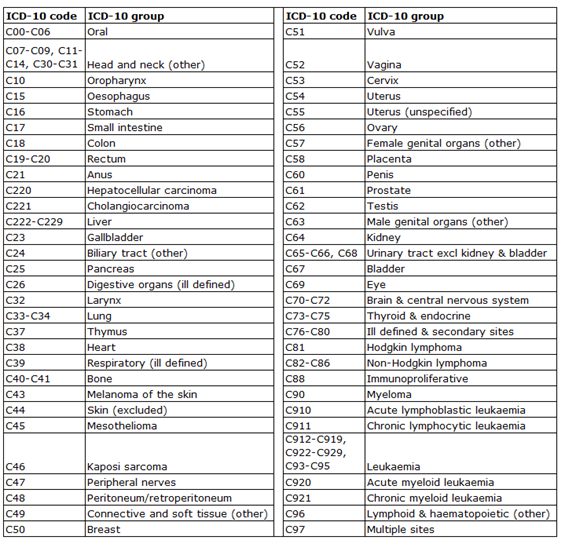
Appendix C
List of cancer groups used for de-duplication of pathology data
Appendix D
Monthly pathology samples vs cancer registration data
Appendix E
2013 European Standard Population
Appendix F
A list of the broad cause groups and corresponding ICD-10 codes
